Understanding and predicting animal dispersal has been challenging because dispersal is a highly plastic trait that depends on multiple ecological factors (Fig. 1). Importantly, dispersal varies between individuals, as not all individuals in a population disperse. Physiology can provide a causal link for individual patterns of dispersal because of the relationship between an animal’s physiological capacity for locomotion and movement propensity (intrinsic state). However, most experimental studies do not directly look at the relationship between physiological capacity and dispersal, but instead investigate dispersal ‘potential’ or ‘tendency’ such as activity level or exploration. Whether the relationship between physiological capacity and dispersal tendency translated to dispersal was the core subject of this meta-analysis.
A second important aspect of physiology-dependent dispersal is that selection for dispersal-prone phenotypes can lead to phenotypic shifts for populations at the range expansion front compared with populations at the stationary centre (Fig. 1). Likewise, populations at the expansion edge are subjected to novel environments acting on their life‐history and dispersal traits. In this study, published in Communications Biology, we conducted a meta-analysis to examine 1) if individual differences in physiological capacity predict dispersal tendency and dispersal, and 2) if populations at the expansion front differ in physiology compared to populations from their range core.

We found most experimental studies investigating 'dispersal tendency' such as activity levels and exploration focused on physiological traits around energy metabolism and locomotor capacity, while studies on dispersal were few (understandably difficult), mainly focused on body condition. This provides important context on the current-state-of knowledge and research gaps for future studies to focus on. On contrary to the current paradigm on physiology-mediated movement, resting and maximal metabolic rate did not predict movement. In the context of dispersal, we proposed that the link between metabolic rate and movement may instead be driven by quality and availability of resources and stored energy. Details on how this may work is discussed in the paper.
For the range expansion analysis, we found populations at their expansion edge had higher thermal tolerance, metabolism, immunity, and hormone levels (Fig. 2a). Differences in physiology between core and edge may therefore have implications for gene flow and adaptation. If physiology was determined genetically, then individuals with particular physiological phenotypes that are likely to disperse would lead to genetic divergence between core and edge populations. Likewise, plasticity may play a role in dispersal, where individuals with greater capacity for plasticity may be more successful at dispersing so that there is a gradient in capacity for plasticity between core and edge. This would be an interesting hypothesis to explore for future studies.

Differences in physiological phenotypes between core-edge populations increased with time since separation, which suggests transgenerational epigenetic effects or adaptation is driving these phenotypic changes if selection plays a role. Interestingly, we noticed a relationship between the rate of dispersal and the temperature of their new environment, where animals moving to warmer areas have higher rates of dispersal. It is not clear whether there is a cause-and-effect relationship that can explain this trend, but if it were robust — and considering the geographical bias in the data — climate change could disrupt dispersal patterns if warmer environments exceeded their heat tolerances.
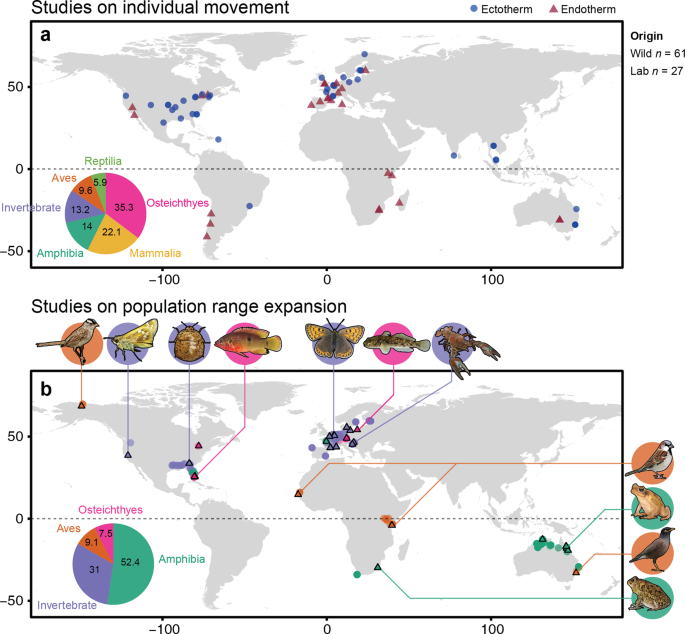
We also revealed strong geographical bias in studies, where most focused on animals in North America and Europe (Fig. 3a), and Australia for core-edge comparisons (Fig 3b). These biases, including study bias on specific physiological traits and taxonomic groups, are unlikely to represent true biological generalities. Clearly, future work needs to focus on broadening the scientific base, particularly because the geographical areas worst affected by climate change are also the least sampled.

Outstanding questions
- Whether physiological differences between the core and edge of distributions are the cause or effect of dispersal;
- Whether physiological differences are mediated genetically or reflect plasticity;
- How will these dynamics affect population structures.
Link to paper: https://doi.org/10.1038/s42003-022-03055-y
Follow Nicholas on Twitter for more research: @NicholasWuNZ
Poster image credit: Vitalij Cerepok, Getty Images






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in